Qing lab in Medical Research Institute Identifies Therapeutic Vulnerabilities for Precision Treatment of Myc-addicted Human Cancers
On October 23, 2016, Dr. Guoliang Qing and colleagues published their latest research entitled “Polo-like Kinase-1 Regulates Myc Stabilization and Activates a Feed-Forward Circuit Promoting Tumor Cell Survival” in Molecular Cell.
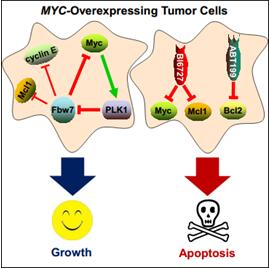
Therapeutic regimens for the high proportion of human cancers driven by Myc are limited. Using MYCN-amplified neuroblastomas and small cell lung carcinomas as model systems, Dr. Qing and colleagues identify that Polo-like Kinase-1 (PLK1) and Myc create a positive interconnected autoregulatory circuit in MYC-overexpressing tumor cells, which is essential for sustaining mutual, high expression. PLK1 specifically binds to the SCF-Fbw7 ubiquitin ligase, phosphorylates it, and promotes its auto-polyubiquitination and proteasomal degradation, counteracting Fbw7-mediated degradation of N-Myc. Stabilized Myc in turn directly activates PLK1 transcription, constituting a feedforward activation loop that reinforces Myc-regulated oncogenic programs. Inhibitors of PLK1 preferentially induce potent apoptosis of MYC-overexpressing tumor cells, and synergistically potentiate the therapeutic efficacies of Bcl2 antagonists. These findings thus reveal a PLK1-Fbw7-Myc signaling circuit that underlies neuroblastoma pathogenesis, and validate PLK1 inhibitors as potential Myc-targeting cancer therapeutics. More importantly, these studies suggest that a combination of volasertib (PLK1 inhibitor) with ABT199 (BCL2 inhibitor), two drugs already exhibiting promising therapeutic benefits in clinical trials, should be translated as a strategy to move forward in future patient care, as patients with MYC deregulation are likely to respond.